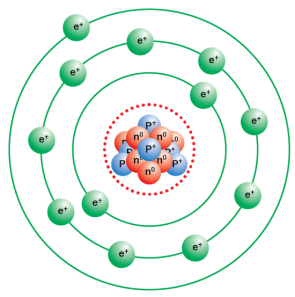
Protons are present in the nucleus of an atom. For the symbol H, D and T tabulate three sub-atomic particles found in each of them.Įlectrons are present outside the nucleus of an atom. Mass number of sulphur = number of neutrons + number of protons = 16 + 16 = 32 Mass number of oxygen = number of neutrons + number of protons = 8+8 =16 With the help of Table 4.1, find out the mass number of oxygen and sulphur atom. 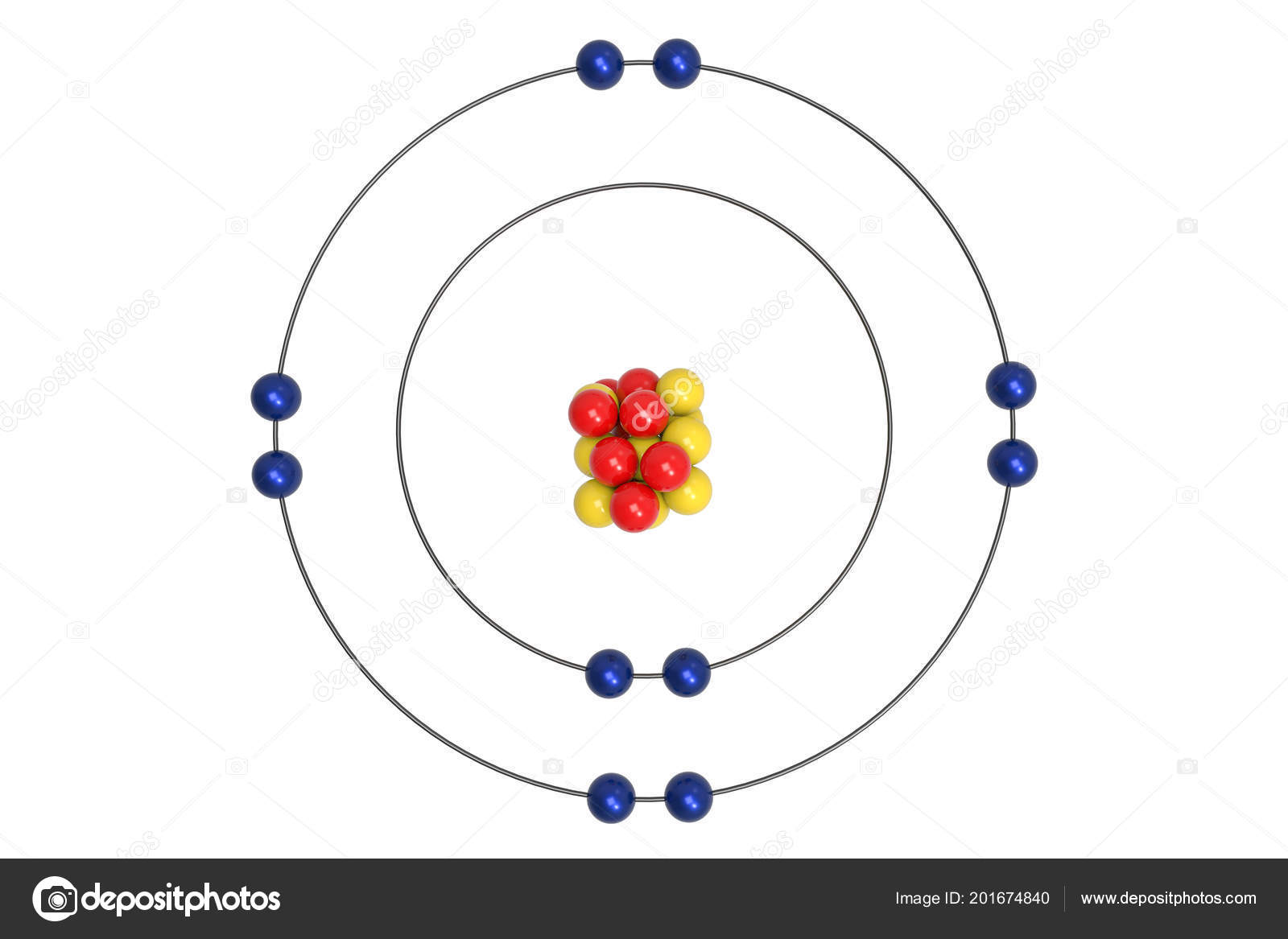
(ii)atom will be neutral (no charge) because number of protons(+) is equal to the number of electrons (-). (i) atomic number = number of protons = number of electrons = 8 (i) what is the atomic number of the atom? and If number of electrons in an atom is 8 and number of protons is also 8, then It becomes easier for magnesium to give away its two valence than to acquire 6 more therefore its valency is two. So the number of valence electrons for sulphur is 6 and it needs 2 more electrons to complete its octet (8). So the number of valence electrons for chlorine is 7 and it needs 1 more electron to complete its octet (8). The atomic number of chlorine is 17 = 2,8,7 Those electrons determine the valency of that atom. The electrons present in the outermost shell of an atom are known as the valence electrons. How will you find the valency of chlorine, sulphur and magnesium? Solution : If K and L shells of an atom are full, then the total number of electron in the atom will be 10 because K shell can accommodate total 2 and L shell can accommodate maximum 8 electrons that makes a total of 10. If K and L shells of an atom are full, then what would be the total number of electrons in the atom? Write the distribution of electrons in carbon and sodium atoms.ĭistribution of electrons in carbon atom: atomic number of carbon = 6 = 2,4ĭistribution of electrons in sodium atom: atomic number of sodium = 11 = 2,8,1 The number of neutrons present in a helium atom Helium atom has an atomic mass of 4 u and two protons in its nucleus. Name the three sub-atomic particles of an atom. Solution : Yes the observations would have been different if the α-particle scattering experiment is carried out using a foil of a metal other than gold. What do you think would be the observation if the α-particle scattering experiment is carried out using a foil of a metal other than gold? Draw a sketch of Bohr’s model of an atom with three shells. Solution : The sub atomic particle proton is present in the nucleus of an atom according to Rutherford’s model of an atom. On the basis of Rutherford’s model of an atom, which subatomic particle is present in the nucleus of an atom? 
Hence the + and – charges are neutralized by each other that makes atom neutral as a whole. Solution : As per Thomson’s model of an atom, the number of electrons (negatively charged) are equal to the number of protons (positively charged) in an atom. On the basis of Thomson’s model of an atom, explain how the atom is neutral as a whole. Solution : That atom will not contain any charge because one negative charge of single electron and one positive charge of single proton are neutralized by each other. If an atom contains one electron and one proton, will it carry any charge or not? Goldstein discovered positively charged rays, those rays are called canal rays. Also read the theory of this Chapter before going to exam. use as a reference of the following NCERT solutions of chapter 4 prepared by Physics Wallah. Get solutions of all chapters of NCERT class 9 Science from Physics Wallah. NCERT Solutions for class 9 Science chapter 4 Structure of Atom is prepared and uploaded for reference by academic team of expert members of Physics Wallah. NCERT Solutions for class 9 Science chapter-4 Structure of Atom
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
